|
As you peruse these examples, notice the mass-balance of the nuclear equations (both mass number and atomic number). One example of electron capture involves beryllium-7. Radioactive Decay Processes There are various types of decay processes that radioactive (unstable) nuclei may undergo to increase their stability. Often the nucleus exists in an excited state as well and emits a gamma ray as it transitions to the ground state of the new nuclide. Electron capture is the primary decay mode for isotopes with a relative. There are four different types of emissions that occur. Electron capture is an example of weak interaction, one of the four fundamental forces. Learning about these forms of nuclear decay is a crucial part of any nuclear physics course. In most instances, the atom changes its identity to become a new element. 00:03 12:50 Brought to you by Sciencing There are three types of radioactive decay: alpha decay, beta decay and gamma decay, although beta decay in itself comes in three different types. Nuclear Decay Processes Radioactive decay involves the emission of a particle and/or energy as one atom changes into another. As a result, a proton transforms into a neutron, and a neutrino is. Example 11.2.1 shows how we can identify a nuclide by balancing the nuclear reaction. For example, after undergoing electron capture, an atom of carbon (with 6 protons) becomes an atom of boron (with 5 protons). (We pronounce the name as "oh zhay", not "aw ger). Electron capture happens when an unstable nucleus absorbs an inner orbital electron. Since an atom loses a proton during electron capture, it changes from one element to another. We call this ejected electron an Auger electron after one of its discoverers, Pierre Victor Auger. 
All it does is carry away excess energy.Īnother electron may absorb excess energy and leave the atom. Many people omit the neutrino in the equation, because it has no mass or charge. The excess energy leaves as an X-ray photon and a neutrino. The inner shell is missing an electron.Īn outer shell electron then drops to a lower energy level to replace the missing electron. The atom stays neutral in charge, but it now exists in an excited state. In the process, a proton combines with the electron and forms a neutron, and an X-ray is released in the process. Confirming a decades-old prediction, scientists found the first clear example of a type of stellar explosion called an electron-capture supernova (large white dot at right). Explanation: Electron capture occurs when the nucleus of an unstable isotope captures an inner-orbital electron. 
The captured electron often comes from the K-shell because those electrons are closest to the nucleus. Electron capture is a process in which a parent nucleus captures one of its orbital electrons and emits a neutrino. During electron capture, an electron in an atoms inner shell is drawn into the nucleus where it combines with a proton, forming a neutron and a neutrino. K- electron capture is the capture of a 1s electron by the nucleus of an unstable isotope. Since a proton becomes a neutron, the number of protons decreases by 1, but the atomic mass stays the same. then((img: Electron.NativeImage) => ).The electron combines with a proton to make a neutron and emits an electron neutrino.Įlectron capture is a major decay mode for isotopes with too many protons in the nucleus. 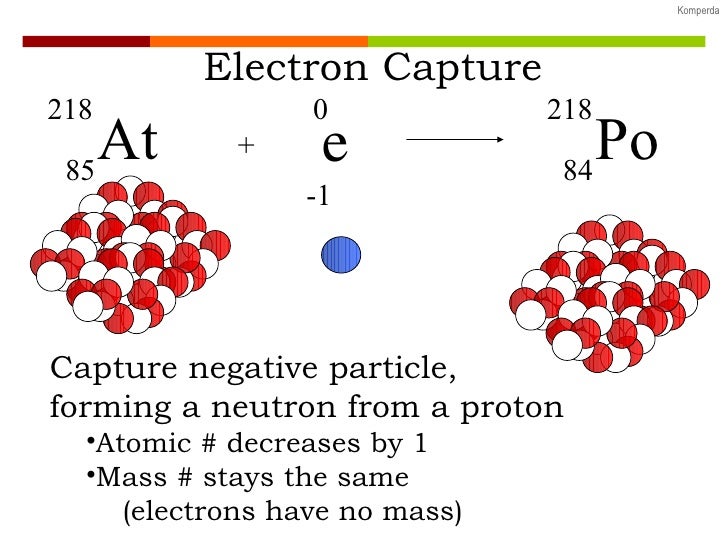
I tried option 1, which is to use capturePage(): ().webContents.capturePage() 
I am getting stuck on the first step, which is the screenshot capturing process. So, when there is an empty space in the inner shells. process this image by sending the image as a byte stream to my python server, and Electron capture is accompanied by the emission of an X ray as the X ray emits gamma radiation.take a screenshot of the entire screen (not the overlay itself which is transparent, but the screen 'underneath'),.I am using Electron to create a Windows application that creates a fullscreen transparent overlay window.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
